Background:
Midostaurin, a multikinase inhibitor that directly inhibits FLT3, is approved for the treatment of adult patients (pts) with newly diagnosed, FLT3-mutated AML. In the phase 3 RATIFY study (NCT00651261), younger adults (aged ≤ 60 y) who received midostaurin plus standard chemotherapy (CT) had significant improvements in survival compared with those that received placebo plus standard CT. To further evaluate the safety and efficacy of midostaurin in FLT3-mutated AML, a phase 3b (NCT03379727) study was initiated that allowed enrollment of younger and older (aged > 60 y) pts and variations in CT regimens, including idarubicin or daunorubicin for 7+3 or 5+2.
Methods:
This is an open-label, single-arm, multicenter study in adults fit for CT with newly diagnosed AML, ECOG performance status (PS) ≤ 2, and a documented FLT3 ITD or TKD mutation. Pts must start their first induction cycle with 7+3 (cytarabine [Ara-C] 100-200 mg/m2/d on days 1-7 + daunorubicin 60-90 mg/m2/d or idarubicin 12 mg/m2/d on days 1-3) or 5+2 (a reduced-dose regimen with these agents) per investigator's discretion and enroll by day 7 of the first induction cycle. Pts are assigned to the 7+3 group if their Ara-C duration is ≥ 7 days, independent of daunorubicin/idarubicin duration, and to the 5+2 group in other cases. Pts may not switch once started on 7+3 or 5+2. Pts receive Ara-C consolidation (dose per investigator's choice). Midostaurin 50 mg bid is administered on days 8-28 of each 28-day induction/consolidation cycle and daily for ≤ 12 cycles of maintenance. Pts are discontinued from the study if not in either complete remission (CR) or CR with incomplete hematologic recovery (CRi) at the end of induction, relapse during consolidation/maintenance, receive a stem cell transplant (SCT), or experience toxicities leading to discontinuation. The primary and secondary endpoints are safety and the proportion of pts achieving CR/CRi, respectively.
Results:
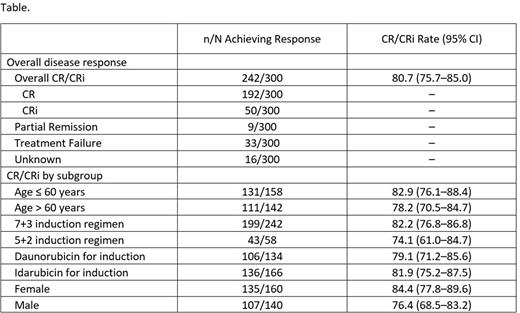
Pts were recruited throughout Europe; study enrollment was closed January 28th, 2020, with 318 pts screened and 17 screen failures. Safety and efficacy analyses focused on 301 pts who received treatment and 300 pts who met all study criteria, respectively. The median age (range) was 59 (19-85) y, and 47.2% were aged > 60 y. Most pts had a FLT3-ITD mutation (82.7%), with 17.6% having a FLT3-TKD mutation. At data cut-off on March 31st, 2020, 63 pts were still receiving treatment. All 301 pts entered induction, 69% entered consolidation, and 26% entered maintenance; 28.7% underwent SCT. The majority of pts (80.4%) were treated with midostaurin plus 7+3; 19.6% received 5+2. Approximately 55.1% received idarubicin and 44.9% received daunorubicin for induction. Pts who received daunorubicin for induction were younger (62.2% vs 45.2% were aged < 60 y) and had a lower frequency of ECOG PS=2 (10.4% vs 18.1%) vs those who received idarubicin. Overall, 242 pts achieved CR/CRi (80.7%). The CR/CRi rates in older vs younger pts, female vs male pts, and pts who received daunorubicin vs idarubicin for induction were similar (Table). The majority of SAEs occurred during induction (75, 24.9%) and consolidation (64, 30.6%); there were 5 (6.3%) SAEs during maintenance. The most common AEs were pyrexia, nausea, diarrhea, and febrile neutropenia. Compared with younger pts, older pts had a higher frequency of grade ≥ 3 AEs (78.6% vs 90.8%) and SAEs (35.8% vs 51.4%), AEs leading to dose adjustment/interruption (30.2% vs 43%), and treatment-related AEs (73.6% vs 78.2%), SAEs (15.7% vs 23.9%), and those leading to discontinuation (6.9% vs 11.3%). Compared with younger pts, older pts had a higher frequency of certain grade ≥3 AEs, including severe infections (31.4% vs 45.1%), leukopenia (40.9% vs 49.3%), QT prolongation (3.1% vs 7.7%) , and pulmonary toxicity (3.8% vs 7.7%). There were 30 deaths (10%) reported during the study (7 younger pts, 23 older pts). Of pts who died, 19 were treated with idarubicin (all were older pts) and 11 were treated with daunorubicin for induction.
Conclusions:
Midostaurin plus CT resulted in high response rates regardless of pt age, sex, induction drug, or alternative CT regimen. The majority of pts received a 7+3 regimen regardless of age. The safety profile of midostaurin plus idarubicin or daunorubicin for induction was generally similar. The safety profile in older and younger pts was consistent with what has been previously reported, with no new safety signals identified.
Sierra:Jazz Pharmaceuticals: Research Funding; Pfizer: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Daiichi Sankyo: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Abbvie: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Novartis: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Astellas: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees; Roche: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees; Gilead-Kite: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees. Griškevičius:Novartis: Research Funding. Cluzeau:Astellas: Membership on an entity's Board of Directors or advisory committees; Amgen: Consultancy, Membership on an entity's Board of Directors or advisory committees; Celgene: Membership on an entity's Board of Directors or advisory committees; Abbvie: Membership on an entity's Board of Directors or advisory committees; Jazz Pharma: Consultancy, Membership on an entity's Board of Directors or advisory committees; Roche: Membership on an entity's Board of Directors or advisory committees; Novartis: Membership on an entity's Board of Directors or advisory committees; Agios: Membership on an entity's Board of Directors or advisory committees; Menarini: Consultancy. Luppi:Gilead Sci: Consultancy, Speakers Bureau; Abbvie: Consultancy; Daiichi-Sankyo: Consultancy; MSD: Consultancy; Novartis: Consultancy, Speakers Bureau; Sanofi: Consultancy. Farkas:Abbvie: Honoraria; MSD: Honoraria; Pfizer: Honoraria; Novartis: Honoraria; Amgen: Honoraria; Johnson & Johnson: Honoraria. Bengoudifa:Novartis: Current Employment. Gilotti:Novartis: Current Employment. Hodzic:Novartis: Current Employment. Rambaldi:Roche: Honoraria; MSD: Honoraria; Abbvie: Honoraria; Jazz: Honoraria; Astellas: Honoraria; Omeros: Honoraria; Novartis: Honoraria; Gilead: Honoraria; Bristol-Myers Squibb: Honoraria; Sanofi: Honoraria; Amgen: Honoraria; Pfizer: Honoraria. Venditti:Novartis: Consultancy, Honoraria, Other: TRAVEL, ACCOMMODATIONS, EXPENSES (paid by any for-profit health care company); Jazz: Consultancy, Honoraria, Other: TRAVEL, ACCOMMODATIONS, EXPENSES (paid by any for-profit health care company); Amgen: Consultancy, Honoraria, Other: TRAVEL, ACCOMMODATIONS, EXPENSES (paid by any for-profit health care company); Pfizer: Consultancy, Honoraria, Other: TRAVEL, ACCOMMODATIONS, EXPENSES (paid by any for-profit health care company), Speakers Bureau; AbbVie: Consultancy, Honoraria, Other: TRAVEL, ACCOMODATIONS, EXPENSES (paid by any for-profit health care company); Janssen: Consultancy, Honoraria, Other: TRAVEL, ACCOMODATIONS, EXPENSES (paid by any for-profit health care company).
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal